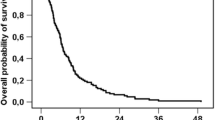
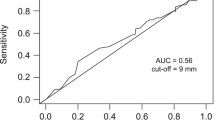
Abstract
Objectives
Pre-therapeutic prediction of outcome is important for clinicians and patients in determining whether selective internal radiation therapy (SIRT) is indicated for hepatic metastases of colorectal cancer (CRC).
Methods
Pre-therapeutic characteristics of 100 patients with colorectal liver metastases (CRLM) treated by radioembolization were analyzed to develop a nomogram for predicting survival. Prognostic factors were selected by univariate Cox regression analysis and subsequent tested by multivariate analysis for predicting patient survival. The nomogram was validated with reference to an external patient cohort (n = 25) from the Bonn University Department of Nuclear Medicine.
Results
Of the 13 parameters tested, four were independently associated with reduced patient survival in multivariate analysis. These parameters included no liver surgery before SIRT (HR:1.81, p = 0.014), CEA serum level ≥ 150 ng/ml (HR:2.08, p = 0.001), transaminase toxicity level ≥2.5× upper limit of normal (HR:2.82, p = 0.001), and summed computed tomography (CT) size of the largest two liver lesions ≥10 cm (HR:2.31, p < 0.001). The area under the receiver-operating characteristic curve for our prediction model was 0.83 for the external patient cohort, indicating superior performance of our multivariate model compared to a model ignoring covariates.
Conclusions
The nomogram developed in our study entailing four pre-therapeutic parameters gives good prediction of patient survival post SIRT.
Key Points
• Four individual parameters predicted reduced survival following SIRT in CRC.
• These parameters were combined into a nomogram of pre-therapeutic risk stratification.
• The model provided good prediction of survival in two independent patient cohorts.

Similar content being viewed by others
References
Donadon M, Ribero D, Morris-Stiff G, Abdalla EK, Vauthey JN (2007) New paradigm in the management of liver-only metastases from colorectal cancer. Gastrointest Cancer Res 1:20–27
Jakobs TF, Hoffmann RT, Poepperl G et al (2007) Mid-term results in otherwise treatment refractory primary or secondary liver confined tumours treated with selective internal radiation therapy (SIRT) using (90)Yttrium resin-microspheres. Eur Radiol 17:1320–1330
Kennedy A, Nag S, Salem R et al (2007) Recommendations for radioembolization of hepatic malignancies using yttrium-90 microsphere brachytherapy: a consensus panel report from the radioembolization brachytherapy oncology consortium. Int J Radiat Oncol Biol Phys 68:13–23
Kennedy AS, Coldwell D, Nutting C et al (2006) Resin 90Y-microsphere brachytherapy for unresectable colorectal liver metastases: modern USA experience. Int J Radiat Oncol Biol Phys 65:412–425
Stubbs RS, O'Brien I, Correia MM (2006) Selective internal radiation therapy with 90Y microspheres for colorectal liver metastases: single-centre experience with 100 patients. ANZ J Surg 76:696–703
Sato KT, Lewandowski RJ, Mulcahy MF et al (2008) Unresectable chemorefractory liver metastases: radioembolization with 90Y microspheres—safety, efficacy, and survival. Radiology 247:507–515
Sangro B, Gil-Alzugaray B, Rodriguez J et al (2008) Liver disease induced by radioembolization of liver tumors: description and possible risk factors. Cancer 112:1538–1546
Kennedy AS, McNeillie P, Dezarn WA et al (2009) Treatment parameters and outcome in 680 treatments of internal radiation with resin 90Y-microspheres for unresectable hepatic tumors. Int J Radiat Oncol Biol Phys 74:1494–1500
Karnofsky DA, Burchenal JH (1949) The clinical evaluation of chemotherapeutic agents in cancer. In: MacLeod CM (ed) Evaluation of chemotherapeutic agents. Columbia Univ Press. Page 196
Goin JE, Salem R, Carr BI et al (2005) Treatment of unresectable hepatocellular carcinoma with intrahepatic yttrium 90 microspheres: factors associated with liver toxicities. J Vasc Interv Radiol 16:205–213
Ho S, Lau WY, Leung TW, Chan M, Johnson PJ, Li AK (1997) Clinical evaluation of the partition model for estimating radiation doses from yttrium-90 microspheres in the treatment of hepatic cancer. Eur J Nucl Med 24:293–298
Boellaard R, O'Doherty MJ, Weber WA et al (2010) FDG PET and PET/CT: EANM procedure guidelines for tumour PET imaging: version 1.0. Eur J Nucl Med Mol Imaging 37:181–200
Fendler WP, Philippe Tiega DB, Ilhan H et al (2013) Validation of several SUV-based parameters derived from 18F-FDG PET for prediction of survival after SIRT of hepatic metastases from colorectal cancer. J Nucl Med 54:1202–1208
Iasonos A, Schrag D, Raj GV, Panageas KS (2008) How to build and interpret a nomogram for cancer prognosis. J Clin Oncol 26:1364–1370
Kanas GP, Taylor A, Primrose JN et al (2012) Survival after liver resection in metastatic colorectal cancer: review and meta-analysis of prognostic factors. Clin Epidemiol 4:283–301
Giacchetti S, Itzhaki M, Gruia G et al (1999) Long-term survival of patients with unresectable colorectal cancer liver metastases following infusional chemotherapy with 5-fluorouracil, leucovorin, oxaliplatin and surgery. Ann Oncol 10:663–669
Morris EJ, Forman D, Thomas JD et al (2010) Surgical management and outcomes of colorectal cancer liver metastases. Br J Surg 97:1110–1118
Bester L, Feitelson S, Milner B, Chua TC, Morris DL (2013) Impact of prior hepatectomy on the safety and efficacy of radioembolization with Yttrium-90 microspheres for patients with unresectable liver tumors. Am J Clin Oncol
Shindoh J, Loyer EM, Kopetz S et al (2012) Optimal morphologic response to preoperative chemotherapy: an alternate outcome end point before resection of hepatic colorectal metastases. J Clin Oncol 30:4566–4572
Veltri A, Guarnieri T, Gazzera C et al (2012) Long-term outcome of radiofrequency thermal ablation (RFA) of liver metastases from colorectal cancer (CRC): size as the leading prognostic factor for survival. Radiol Med 117:1139–1151
Nordlinger B, Guiguet M, Vaillant JC et al (1996) Surgical resection of colorectal carcinoma metastases to the liver. A prognostic scoring system to improve case selection, based on 1568 patients. Association Francaise de Chirurgie. Cancer 77:1254–1262
Fong Y, Fortner J, Sun RL, Brennan MF, Blumgart LH (1999) Clinical score for predicting recurrence after hepatic resection for metastatic colorectal cancer: analysis of 1001 consecutive cases. Ann Surg 230:309–318, discussion 318–321
Nagashima I, Takada T, Matsuda K et al (2004) A new scoring system to classify patients with colorectal liver metastases: proposal of criteria to select candidates for hepatic resection. J Hepato-Biliary-Pancreat Surg 11:79–83
Rees M, Tekkis PP, Welsh FK, O'Rourke T, John TG (2008) Evaluation of long-term survival after hepatic resection for metastatic colorectal cancer: a multifactorial model of 929 patients. Ann Surg 247:125–135
Berber E, Pelley R, Siperstein AE (2005) Predictors of survival after radiofrequency thermal ablation of colorectal cancer metastases to the liver: a prospective study. J Clin Oncol 23:1358–1364
Ohlsson B, Stenram U, Tranberg KG (1998) Resection of colorectal liver metastases: 25-year experience. World J Surg 22:268–276, discussion 276–267
Schindl M, Wigmore SJ, Currie EJ, Laengle F, Garden OJ (2005) Prognostic scoring in colorectal cancer liver metastases: development and validation. Arch Surg 140:183–189
Konopke R, Kersting S, Distler M et al (2009) Prognostic factors and evaluation of a clinical score for predicting survival after resection of colorectal liver metastases. Liver Int 29:89–102
Rougier P, Milan C, Lazorthes F et al (1995) Prospective study of prognostic factors in patients with unresected hepatic metastases from colorectal cancer. Fondation Francaise de Cancerologie Digestive. Br J Surg 82:1397–1400
Stangl R, Altendorf-Hofmann A, Charnley RM, Scheele J (1994) Factors influencing the natural history of colorectal liver metastases. Lancet 343:1405–1410
Schmoll HJ, Van Cutsem E, Stein A et al (2012) ESMO Consensus Guidelines for management of patients with colon and rectal cancer. A personalized approach to clinical decision making. Ann Oncol 23:2479–2516
Excellence NIfHaC (2011) The diagnosis and management of colorectal cancer CG131. National Institute for Health and Care Excellence, London
Acknowledgments
We thank Dr. Alexander Crispin (Institute of Medical Informatics, Biometry, and Epidemiology, Munich, Germany) for valuable statistical advice. The authors acknowledge professional manuscript revisions by Inglewood Biomedical Editing. The scientific guarantor of this publication is Wolfgang Peter Fendler, MD. The authors of this manuscript declare relationships with the following companies: A.H., T.J., P.B., and S.E. have received fees or travel-related expense reimbursement for oral presentations at events organized by SIRTeX Medical (Sydney, Australia). P.P. and T.J. have an advisory relationship with SIRTeX Medical. The other authors declare that they have no potential conflicts of interest. The authors state that this work has not received any funding. One of the contributors, Dr. Alexander Crispin (Institute of Medical Informatics, Biometry, and Epidemiology, Munich, Germany), has significant statistical expertise. Institutional review board approval was obtained. Written informed consent was waived by the institutional review board. Some study subjects or cohorts have been previously reported in: Fendler WP, Philippe Tiega DB, Ilhan H, et al. Validation of Several SUV-Based Parameters Derived from 18F-FDG PET for Prediction of Survival after SIRT of Hepatic Metastases from Colorectal Cancer. J Nucl Med. 2013;54:1202–1208. Methodology: retrospective, diagnostic or prognostic multi-centre study.
Author information
Authors and Affiliations
Corresponding author
Electronic supplementary material
Below is the link to the electronic supplementary material.
Supplemental Fig. S1
Diagram showing the flow of patients through the study (JPEG 1280 kb)
Supplemental Fig. S2
Underlying statistic model for building the nomogram. The probability of 1-year survival is calculated as follows: Probability of 1-year survival = EXP(-CBH*EXP(a*total points)); CBH = 0.215; EXP = Exponential function; CBH = Cumulative baseline hazard (JPEG 168 kb)
Supplemental Fig. S3
Receiver-operating-characteristic curves of the prediction model versus a model that ignores covariates for (A) internal and (B) external validation (JPEG 603 kb)
Rights and permissions
About this article
Cite this article
Fendler, W.P., Ilhan, H., Paprottka, P.M. et al. Nomogram including pretherapeutic parameters for prediction of survival after SIRT of hepatic metastases from colorectal cancer. Eur Radiol 25, 2693–2700 (2015). https://doi.org/10.1007/s00330-015-3658-7
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00330-015-3658-7